



VETERINARY INJECTION MANUFACTURER IN SIKKIM
Product Details:
- Shelf Life 24 months from manufacturing date (subject to formulation)
- Indication For prevention and treatment of infections, inflammations, or specific animal health conditions
- Packaging Type Glass vials, ampoules, or plastic containerssealed and sterile
- Medicine Type Veterinary Injection
- Physical Form Liquid
- Ingredients As per composition requirements (customizable on request)
- Treatments & Functions Used for various animal health therapeutic purposes as per veterinary prescription
- Click to View more
X
VETERINARY INJECTION MANUFACTURER IN SIKKIM Price And Quantity
- 1000 Carton
- Available for export with regulatory documentation
- Intramuscular/Intravenous/Subcutaneous (as per product specifications)
- Customizable per export or domestic requirements
- Sikkim, India (Certified pharmaceutical zone)
- Autoclaved, sterile fill finish processes
- cGMP compliant, WHO-GMP certified plant
- As per agreement or inquiry
- DMF/CTD dossiers provided on request
- Third party/private label manufacturing available
- Available on bulk order
- Stringent batch release analytics and documentation
VETERINARY INJECTION MANUFACTURER IN SIKKIM Product Specifications
- Veterinary Injection
- As per composition requirements (customizable on request)
- For prevention and treatment of infections, inflammations, or specific animal health conditions
- Glass vials, ampoules, or plastic containerssealed and sterile
- Used for various animal health therapeutic purposes as per veterinary prescription
- Keep in a cool and dry place. Protect from sunlight.
- Specification based on formulation; e.g., 1 ml, 5 ml, or as directed by the veterinarian
- Vial/ampoule packaging; typically 10 ml, 30 ml, or as specified
- 24 months from manufacturing date (subject to formulation)
- Liquid
- Available for export with regulatory documentation
- Intramuscular/Intravenous/Subcutaneous (as per product specifications)
- Customizable per export or domestic requirements
- Sikkim, India (Certified pharmaceutical zone)
- Autoclaved, sterile fill finish processes
- cGMP compliant, WHO-GMP certified plant
- As per agreement or inquiry
- DMF/CTD dossiers provided on request
- Third party/private label manufacturing available
- Available on bulk order
- Stringent batch release analytics and documentation
Product Description
WE PROVIDE VETERINARY PCD FRANCHISE , VETERINARY THIRD PARTY MANUFACTURING , VETERINARY INJECTION MANUFACTURER, VETERINARY BOLUS MANUFACTURER, VETRINARY ANIMAL FEED SUPPLEMENT, OXYTETRACYCLINE INJECTION, TOP TEN VETERINARY COMPANY,
VETERINARY INJECTION
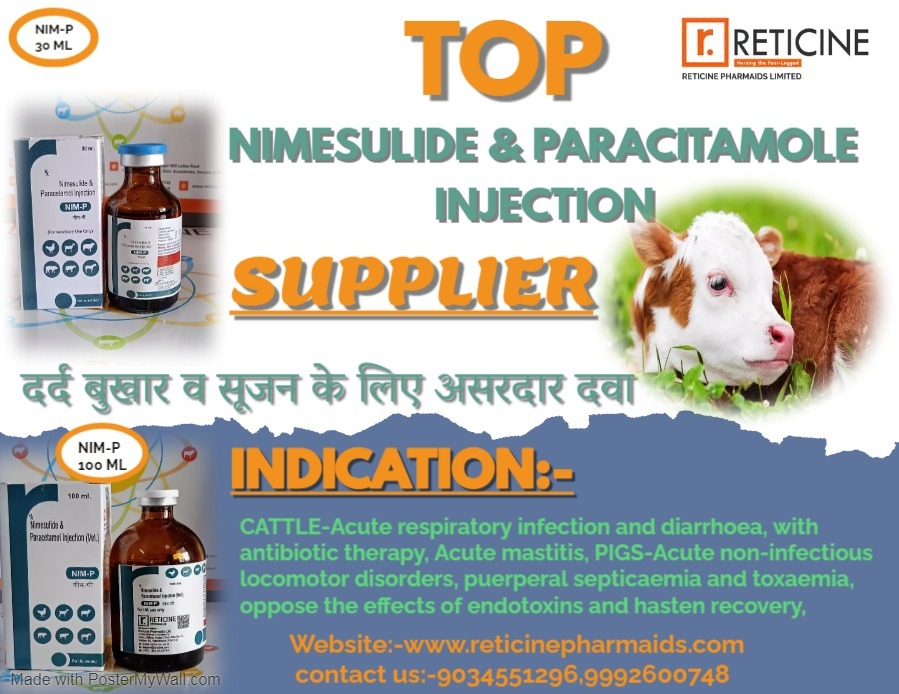
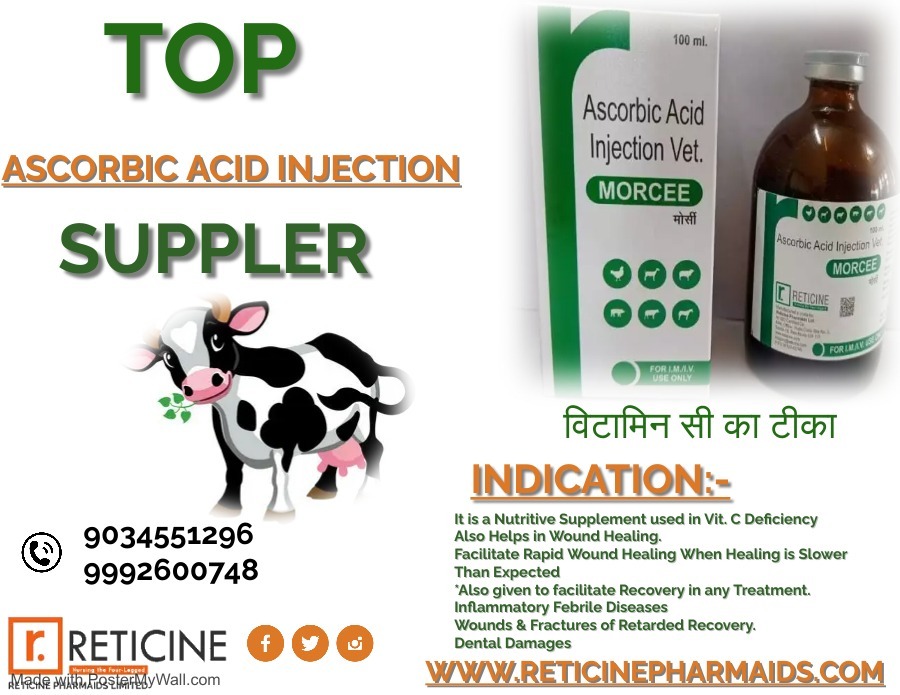
MELOXICAM, MELOXICAM PARACETAMOL, NIMESULIDE, NIMESULIDE PARACETAMOL, OXYTETRACYCLINE, PIROXICAM PARACETAMOL, IVERMECTION, GENTAMICIN, FLUNIXIN MEGLUMINE, ENROFLOXACIN ETC.
DRY POWDER INJECTION
CEFTRIAXONE, CEFTRIAXONE SULBACTUM, CEFOPARAZONE SULBACTUM, CEFOPERAZONE TAZOBACTUM, CEFTIOFUR, AMOXYCILLIN AND POTASSIUM CLAVULANATE, AMOXYCILLIN AND CLOXACILLIN ETC.
State-of-the-Art Manufacturing Facility
Located in Sikkims certified pharmaceutical zone, our manufacturing plant operates under rigorous cGMP and WHO-GMP norms, ensuring each batch of veterinary injections is produced safely and responsibly. Sterility is maintained through autoclaving and a specialized sterile fill-finish process, safeguarding animal health and practitioner trust. Advanced quality control analytics accompany every batch, backed by complete documentation.
Comprehensive Customization & Export Services
We offer third-party and private label veterinary injection manufacturing, enabling brands to expand with bespoke solutions. Our export services include essential regulatory documentation, such as DMF/CTD dossiers, tailored to global requirements. Label languages and formulations are customized per client specifications, supporting international distribution and compliance.
FAQs of VETERINARY INJECTION MANUFACTURER IN SIKKIM:
Q: How are veterinary injections manufactured in your Sikkim facility?
A: Our facility in Sikkim strictly follows cGMP and WHO-GMP protocols. Each batch undergoes autoclaving and sterile fill-finish processes, ensuring product integrity and sterility. Advanced analytics and documentation support every release.Q: What types of animals can use these veterinary injections?
A: Our injections are formulated for use in cattle, horses, sheep, goats, dogs, cats, and other livestock, as per veterinary prescription and specific animal health requirements.Q: When can clients request custom veterinary formulations?
A: Clients may request custom formulations for bulk orders at any time. We collaborate to tailor ingredients and dosage strength, meeting unique therapeutic or market requirements.Q: Where can these veterinary injections be exported?
A: We have the capability to export veterinary injections internationally, providing DMF/CTD dossiers and regulatory documentation as required for destination compliance. Packaging and labeling can be customized for each market.Q: What are the administration routes for these injections?
A: Depending on the product specifications, our veterinary injections can be administered intramuscularly, intravenously, or subcutaneously, as prescribed by a licensed veterinarian.Q: How is quality and sterility assured in your manufacturing process?
A: Quality is maintained through stringent sterility controls, including autoclaving and sterile fill-finish operations. Batch release includes robust analytics and comprehensive quality documentation.Q: What storage conditions are recommended for these injections?
A: Veterinary injections should be stored in a cool, dry place, protected from sunlight, to retain efficacy and shelf life, which typically extends 24 months from the manufacturing date.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Other Products in 'VETERINARY.' category
We deal only in bulk quantity orders.












 Send Inquiry
Send Inquiry Send SMS
Send SMS