




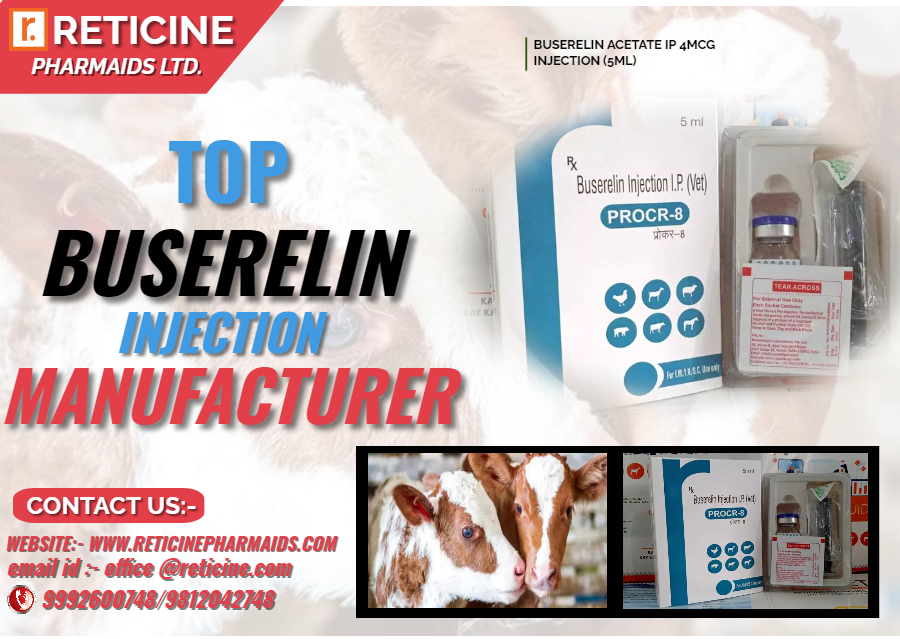
INJECTION MANUFACTURER IN UTTARAKHAND
Product Details:
- Indication Varies with specific injection type
- Shelf Life 2-3 years from the date of manufacture
- Packaging Type Glass Vial, Ampoule, Plastic Vial
- Medicine Type Injection
- Physical Form Liquid
- Ingredients Pharmaceutical API as per formulation
- Treatments & Functions Therapeutic and medicinal purposes as per composition
- Click to View more
INJECTION MANUFACTURER IN UTTARAKHAND Price And Quantity
- 1000 Carton
- Contract manufacturing under client brand
- Sterile Injection
- Provided for domestic and export markets
- As prescribed by healthcare professional
- Available as per formulation or packaging request
- Intravenous, Intramuscular, Subcutaneous
- Uttarakhand, India
- As per client requirement
- Stringent QC and QA processes
INJECTION MANUFACTURER IN UTTARAKHAND Product Specifications
- Varies with specific injection type
- Injection
- Customizable as per client requirement
- Suitable for hospital and clinical use
- 1ml, 2ml, 5ml, 10ml or as specified
- Pharmaceutical API as per formulation
- Store in a cool and dry place
- Therapeutic and medicinal purposes as per composition
- 2-3 years from the date of manufacture
- Liquid
- Glass Vial, Ampoule, Plastic Vial
- Contract manufacturing under client brand
- Sterile Injection
- Provided for domestic and export markets
- As prescribed by healthcare professional
- Available as per formulation or packaging request
- Intravenous, Intramuscular, Subcutaneous
- Uttarakhand, India
- As per client requirement
- Stringent QC and QA processes
Product Description
INJECTIONS
MELOXICAM, MELOXICAM PARACETAMOL, NIMESULIDE, NIMESULIDE PARACETAMOL, OXYTETRACYCLINE, PIROXICAM PARACETAMOL, IVERMECTION, GENTAMICIN, FLUNIXIN MEGLUMINE, ENROFLOXACIN, CLOPROSTENTOL, CHLOROHENIRAMINE MALEATE, BUTAPHOSPHAN CYNOCOBALAMIN, ATROPINE SULPHATE, AMIKACIN SULPHATE, DEXAMETHASONE, HYDROXY PROGESTERONE, ISOFLUPREDONE, KETOPROFEN,LEVOFLOXACIN, DICYCLOMINE, MECOBALAMIN, NATURAL PROGESTERONE, PHENIRAMINE, SODIUM ACID PHOSPHATE, SULPHADIMIDINE,VITAMINA, VITAMIN B COMPLEX, VITAMIN B COMPLEX WITH LIVER EXTRACT(B1,B2,B3,B12), VITAMINB1,B6,B12 ETC.
DRY POWDER INJECTION
CEFTRIAXONE, CEFTRIAXONE SULBACTUM, CEFOPARAZONE SULBACTUM, CEFOPERAZONE TAZOBACTUM, CEFTIOFUR, AMOXYCILLIN AND POTASSIUM CLAVULANATE, AMOXYCILLIN AND CLOXACILLIN, STREPTOPENICILLIN FOR SUSPENSION, AMOXYCILLIN AND CLOXACILLIN, CEFTRIAXONE TAZOBACTUM ETC.
Comprehensive Contract Manufacturing Services
Located in Uttarakhand, our facility offers complete contract manufacturing, supporting both domestic and international clients under their own brands. We provide flexibility in batch size, product composition, and packaging, ensuring all regulatory and clinical needs are met efficiently.
Stringent Quality Assurance and Customization
Every batch undergoes rigorous quality control and assurance, ensuring the highest standards for sterile injections. Clients can request custom formulations or packaging to align with specific medical, regulatory, or branding requirements.
Regulatory and Export Support
From documentation to product registration, our team assists clients in securing all necessary approvals in domestic and global markets. We help streamline the process, making export and compliance straightforward for our partners.
FAQs of INJECTION MANUFACTURER IN UTTARAKHAND:
Q: How do you ensure the quality and sterility of your injectable products?
A: Our manufacturing facility in Uttarakhand adheres to stringent QC and QA processes at every stage, with specialized equipment and controlled environments that maintain sterility and product integrity for all formulations.Q: What is the process for starting contract manufacturing under our brand?
A: We begin with client consultations to define formulation, packaging, and quantity requirements. Once agreed, we offer comprehensive support from sample preparation to production, packaging, and regulatory documentation, including product registration for domestic and export markets.Q: Which types of injections can you manufacture and what customization options are available?
A: We manufacture intravenous, intramuscular, and subcutaneous injections using pharmaceutical APIs as per your specifications. Customization includes dose/strength, packaging type, formulation ingredients, and labeling to suit hospital, clinical, or retail needs.Q: When can clients expect delivery after placing an order?
A: Manufacturing timelines depend on order specifications and regulatory requirements. Typically, dispatch occurs within agreed lead times once product approvals are in place, with support for both domestic distribution and exports.Q: Where are your products manufactured and who do you supply to?
A: All products are manufactured at our advanced facility in Uttarakhand, India. We supply to hospitals, clinics, dealers, exporters, retailers, and wholesalers, both within India and internationally.Q: What are the storage and usage recommendations for your injections?
A: Sterile injections should be stored in a cool, dry place as indicated on the packaging. They must be administered strictly as prescribed by healthcare professionals for their intended therapeutic use.Q: What benefits do clients gain by partnering with your contract manufacturing services?
A: Clients benefit from tailored solutions, rigorous quality assurance, flexible batch sizes, packaging customization, export support, and comprehensive regulatory assistance, ensuring efficient market entry and reliable supply.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+













 Send Inquiry
Send Inquiry Send SMS
Send SMS