




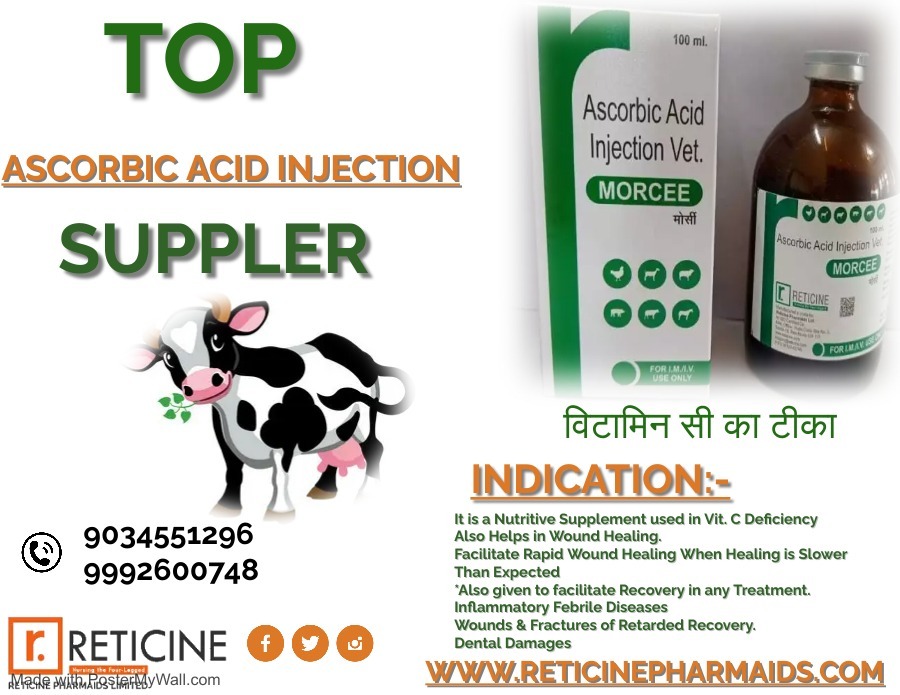
INJECTION MANUFACTURER IN HIMACHAL PRADESH
Product Details:
- Packaging Type Glass Vial / Ampoule
- Indication Treatment as per specific medical condition
- Shelf Life 24 Months from the date of manufacturing
- Medicine Type Injection
- Physical Form Liquid
- Ingredients Pharmaceutical Grade Compounds
- Treatments & Functions Used for therapeutic purposes as prescribed
- Click to View more
INJECTION MANUFACTURER IN HIMACHAL PRADESH Price And Quantity
- 1000 Carton
- Himachal Pradesh, India
- Hospital and Clinical Use
- Available for Domestic & International Supply
- Sterile, Aseptic Processing
- Tested for Purity and Potency
- Sterile Injection for Intravenous or Intramuscular use
- Negotiable as per client requirement
- Regulatory and Dossier Assistance Available
INJECTION MANUFACTURER IN HIMACHAL PRADESH Product Specifications
- Liquid
- Glass Vial / Ampoule
- Store in a cool, dry place, away from direct sunlight
- Varies depending on formulation
- As directed by healthcare professional
- Injection
- Used for therapeutic purposes as prescribed
- Treatment as per specific medical condition
- 24 Months from the date of manufacturing
- Pharmaceutical Grade Compounds
- Provided as per required dosage
- Himachal Pradesh, India
- Hospital and Clinical Use
- Available for Domestic & International Supply
- Sterile, Aseptic Processing
- Tested for Purity and Potency
- Sterile Injection for Intravenous or Intramuscular use
- Negotiable as per client requirement
- Regulatory and Dossier Assistance Available
Product Description
WE PROVIDE VETERINARY PCD FRANCHISE , VETERINARY THIRD PARTY MANUFACTURING , VETERINARY INJECTION MANUFACTURER, VETERINARY BOLUS MANUFACTURER, VETERINARY ANIMAL FEED SUPPLEMENT, OXYTETRACYCLINE INJECTION, TOP TEN VETERINARY COMPANY,
INJECTIONS
MELOXICAM, MELOXICAM PARACETAMOL, NIMESULIDE, NIMESULIDE PARACETAMOL, OXYTETRACYCLINE, PIROXICAM PARACETAMOL, IVERMECTION, GENTAMICIN, FLUNIXIN MEGLUMINE, ENROFLOXACIN, CLOPROSTENTOL, CHLOROHENIRAMINE MALEATE, BUTAPHOSPHAN CYNOCOBALAMIN, ATROPINE SULPHATE, AMIKACIN SULPHATE, DEXAMETHASONE, HYDROXY PROGESTERONE, ISOFLUPREDONE, KETOPROFEN,LEVOFLOXACIN, DICYCLOMINE, MECOBALAMIN, NATURAL PROGESTERONE, PHENIRAMINE, SODIUM ACID PHOSPHATE, SULPHADIMIDINE,VITAMINA, VITAMIN B COMPLEX, VITAMIN B COMPLEX WITH LIVER EXTRACT(B1,B2,B3,B12), VITAMINB1,B6,B12 ETC.
DRY POWDER INJECTION
CEFTRIAXONE, CEFTRIAXONE SULBACTUM, CEFOPARAZONE SULBACTUM, CEFOPERAZONE TAZOBACTUM, CEFTIOFUR, AMOXYCILLIN AND POTASSIUM CLAVULANATE, AMOXYCILLIN AND CLOXACILLIN, STREPTOPENICILLIN FOR SUSPENSION, AMOXYCILLIN AND CLOXACILLIN, CEFTRIAXONE TAZOBACTUM ETC.
Advanced Sterile Injection Manufacturing
We utilize aseptic processing to produce sterile injections tailored for intravenous and intramuscular applications. Our production facility in Himachal Pradesh follows rigorous protocols to maintain product integrity, combining state-of-the-art equipment with advanced technological standards. Each batch is subjected to comprehensive quality assurance testing to guarantee safety and therapeutic effectiveness.
Comprehensive Regulatory & Export Solutions
As a manufacturer with export capabilities, we assist clients with regulatory and dossier documentation, paving the way for smooth registration processes in diverse markets. Our products are supplied across India and internationally, providing solutions for hospitals, clinics, and pharmaceutical distributors. Flexible order volumes accommodate unique business needs.
FAQs of INJECTION MANUFACTURER IN HIMACHAL PRADESH:
Q: How are the sterile injections manufactured at your Himachal Pradesh facility?
A: Injections are produced through aseptic processing using pharmaceutical grade compounds, ensuring sterility and accuracy in formulation. Strict quality control protocols are followed at every step to guarantee purity and efficacy.Q: What is the recommended usage for these sterile injections?
A: They are intended for hospital and clinical use, and should only be administered as directed by qualified healthcare professionals for therapeutic purposes specified in the prescription.Q: What support do you offer for product registration and regulatory compliance?
A: We provide comprehensive assistance with regulatory filings and dossier preparation for both domestic and international supply, helping clients meet necessary compliance requirements.Q: How should the injections be stored for optimal preservation?
A: The injections should be stored in a cool, dry place away from direct sunlight to maintain sterility, potency, and safeguard their shelf life of up to 24 months.Q: Can you accommodate clients with varying order quantities?
A: Yes, minimum order quantity is negotiable as per client requirements, which allows for flexibility in dealing with diverse business volumes including dealers, exporters, and wholesalers.Q: What forms of packaging are available for your injections?
A: Our sterile injections are provided in glass vials or ampoules, designed to prevent contamination and preserve the quality of the formulation throughout its storage and use.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+













 Send Inquiry
Send Inquiry Send SMS
Send SMS