
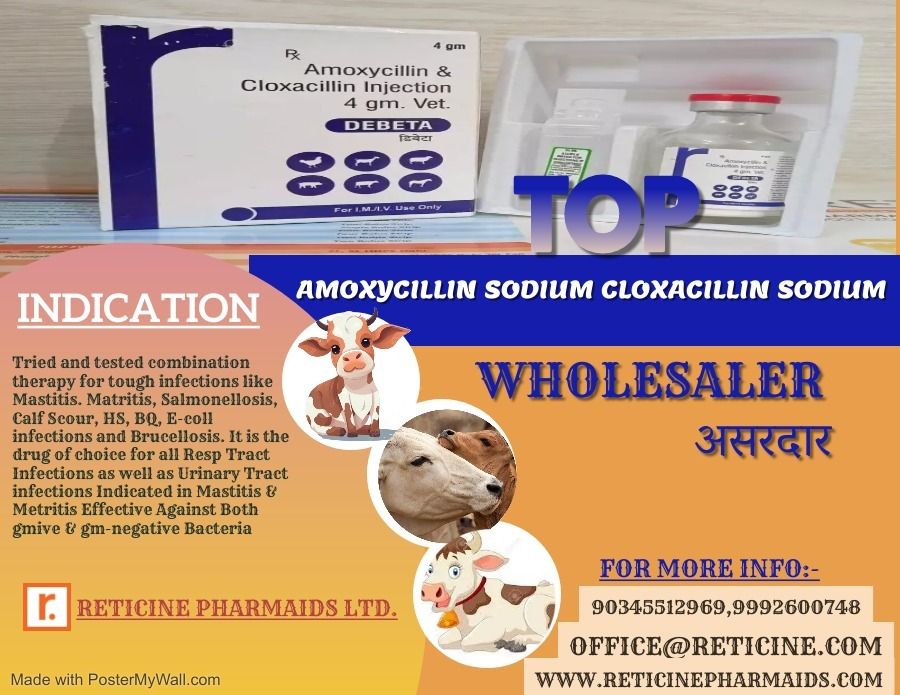




INJECTION MANUFACTURER IN ANDHRA PRADESH
Product Details:
- Indication Varies details provided in product-specific datasheet
- Shelf Life 24 months from date of manufacture
- Packaging Type Ampoules or vials, individually packed or in cartons
- Medicine Type Injection
- Physical Form Liquid
- Ingredients Pharmaceutical grade compounds as per formulation
- Treatments & Functions For the treatment of indicated medical conditions as prescribed
- Click to View more
INJECTION MANUFACTURER IN ANDHRA PRADESH Price And Quantity
- 1000 Carton
- Andhra Pradesh, India
- Complies with Indian Pharmacopoeia (IP) or as specified
- Registered under Indian Drug & Cosmetics Act
- For hospital/institutional use only. Not for retail sale unless permitted.
- Parenteral administration only, as directed by healthcare professional
- Terminally sterilized and pyrogen-free
- Available on each pack
- Shipped in temperature-controlled packaging if required
INJECTION MANUFACTURER IN ANDHRA PRADESH Product Specifications
- Liquid
- Injection
- As specified by product e.g., 1 ml, 2 ml, 5 ml ampoules/vials
- Adults and children as advised by physician
- Pharmaceutical grade compounds as per formulation
- Varies details provided in product-specific datasheet
- Store in a cool, dry place. Protect from light.
- Ampoules or vials, individually packed or in cartons
- 24 months from date of manufacture
- For the treatment of indicated medical conditions as prescribed
- Varies as per product variant (commonly 1 ml, 2 ml, 5 ml, 10 ml)
- Andhra Pradesh, India
- Complies with Indian Pharmacopoeia (IP) or as specified
- Registered under Indian Drug & Cosmetics Act
- For hospital/institutional use only. Not for retail sale unless permitted.
- Parenteral administration only, as directed by healthcare professional
- Terminally sterilized and pyrogen-free
- Available on each pack
- Shipped in temperature-controlled packaging if required
Product Description
INJECTIONS
MELOXICAM, MELOXICAM PARACETAMOL, NIMESULIDE, NIMESULIDE PARACETAMOL, OXYTETRACYCLINE, PIROXICAM PARACETAMOL, IVERMECTION, GENTAMICIN, FLUNIXIN MEGLUMINE, ENROFLOXACIN, CLOPROSTENTOL, CHLOROHENIRAMINE MALEATE, BUTAPHOSPHAN CYNOCOBALAMIN, ATROPINE SULPHATE, AMIKACIN SULPHATE, DEXAMETHASONE, HYDROXY PROGESTERONE, ISOFLUPREDONE, KETOPROFEN,LEVOFLOXACIN, DICYCLOMINE, MECOBALAMIN, NATURAL PROGESTERONE, PHENIRAMINE, SODIUM ACID PHOSPHATE, SULPHADIMIDINE,VITAMINA, VITAMIN B COMPLEX, VITAMIN B COMPLEX WITH LIVER EXTRACT(B1,B2,B3,B12), VITAMINB1,B6,B12 ETC.
DRY POWDER INJECTION
CEFTRIAXONE, CEFTRIAXONE SULBACTUM, CEFOPARAZONE SULBACTUM, CEFOPERAZONE TAZOBACTUM, CEFTIOFUR, AMOXYCILLIN AND POTASSIUM CLAVULANATE, AMOXYCILLIN AND CLOXACILLIN, STREPTOPENICILLIN FOR SUSPENSION, AMOXYCILLIN AND CLOXACILLIN, CEFTRIAXONE TAZOBACTUM ETC.
Advanced Sterilization and Quality Assurance
Our injections are manufactured at a state-of-the-art facility in Andhra Pradesh, utilizing terminal sterilization processes to ensure both sterility and freedom from pyrogens. Rigorous quality protocols guarantee compliance with Indian Pharmacopoeia or specified standards, offering reliability in every product supplied.
Comprehensive Registration and Compliance
Every injectable formulated at our facility is fully registered under the Indian Drug & Cosmetics Act. Detailed labeling, including batch number and manufacturing date, ensures transparency and traceability. We maintain strict adherence to statutory and regulatory requirements for pharmaceutical production.
Custom Packaging and Safe Dispatch
Each product is securely packaged in ampoules or vials, packed individually or in cartons, and dispatched in temperature-controlled conditions if required. Storage and handling guidelines are provided to ensure product stability and efficacy throughout shelf life.
FAQs of INJECTION MANUFACTURER IN ANDHRA PRADESH:
Q: How are your injectable products shipped to maintain quality?
A: Our injectable products are shipped in temperature-controlled packaging when required to preserve their stability and efficacy. We follow strict dispatch protocols to ensure the medicine remains safe and effective during transport.Q: What quality standards do your injections adhere to?
A: All injections are formulated in compliance with Indian Pharmacopoeia (IP) standards or as specified in their approved formulation. Each batch is terminally sterilized, pyrogen-free, and registered under the Indian Drug & Cosmetics Act.Q: When should these injections be administered?
A: These injections should be administered only as directed by a certified healthcare professional in a hospital or institutional setting. Detailed dosing and timing instructions are provided in the product-specific datasheet.Q: Where can your injectable medicines be purchased or used?
A: Our injectable pharmaceuticals are intended exclusively for hospital or institutional use, not for retail sale unless officially permitted. They can be accessed through authorized dealers, wholesalers, or as supplied directly to healthcare facilities.Q: What steps are taken to ensure the safety of each injection?
A: Each injection batch undergoes terminal sterilization and comprehensive quality checks to ensure it is both sterile and pyrogen-free. Every pack includes batch and manufacturing details for traceability and regulatory compliance.Q: What benefits do these injectables offer over other forms?
A: Parenteral administration allows for rapid onset of action and improved bioavailability, making these injectables ideal for situations where quick and precise dosing is required as diagnosed by physicians.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+












 Send Inquiry
Send Inquiry Send SMS
Send SMS